|
Unique properties of lamp2a compared to other lamp2 isoforms. A receptor for the selective uptake and degradation of proteins by lysosomes. An intralysosomal hsp70 is required for a selective pathway of lysosomal protein degradation. Import of a cytosolic protein into lysosomes by chaperone-mediated autophagy depends on its folding state. A molecular chaperone complex at the lysosomal membrane is required for protein translocation. Semin Cell Dev Biol 2010, 21: 719–726.Īgarraberes FA, Dice JF. Chaperone-mediated autophagy: molecular mechanisms and physiological relevance. Mechanisms of chaperone-mediated autophagy. Selective binding and uptake of ribonuclease A and glyceraldehyde-3- phosphate dehydrogenase by isolated rat liver lysosomes. Trends Cell Biol 2012, 22: 407–417.Ĭuervo AM, Terlecky SR, Dice JF, Knecht E. Chaperone–mediated autophagy: a unique way to enter the lysosome world. Chaperone-mediated autophagy: roles in disease and aging. 
Peptide sequences that target cytosolic proteins for lysosomal proteolysis. Chaperone-mediated autophagy: machinery, regulation and biological consequences. Constitutive upregulation of chaperone-mediated autophagy in Huntington's disease. Koga H, Martinez-Vicente M, Arias E, Kaushik S, Sulzer D, Cuervo AM. Chaperone-mediated autophagy markers in Parkinson disease brains. J Cell Biol 2009, 187: 1083–1099.Īlvarez-Erviti L, Rodriguez-Oroz MC, Cooper JM, Caballero C, Ferrer I, Obeso JA, et al. IKK phosphorylates Huntingtin and targets it for degradation by the proteasome and lysosome. Thompson LM, Aiken CT, Kaltenbach LS, Agrawal N, Illes K, Khoshnan A, et al. Chaperone-mediated autophagy in health and disease. Impaired degradation of mutant a-synuclein by chaperonemediated autophagy. Landes Bioscience Austin, TX, 2000.Ĭuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D. Lysosomal pathways of protein degradation. Autophagic tubes vacuolar invaginations involved in lateral membrane sorting and inverse vesicle budding. Müller O, Sattler T, Flötenmeyer M, Schwarz H, Plattner H, Mayer A. Regulation of microautophagy and basal protein turnover in rat liver. Uptake-microautophagy-and degradation of exogenous proteins by isolated rat liver lysosomes: effects of pH, ATP, and inhibitors of proteolysis. Autophagosome formation in mammalian cells. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Autophagy as a regulated pathway of cellular degradation. Overall, the findings provide evidence that diabetes promotes retinal REDD1 protein content via a post-transcriptional effect and potentially identifies superior therapeutic targets for preventing the pathological increase in REDD1.Klionsky DJ, Emr SD. Additional mechanistic details of this new regulatory pathway will be presented. 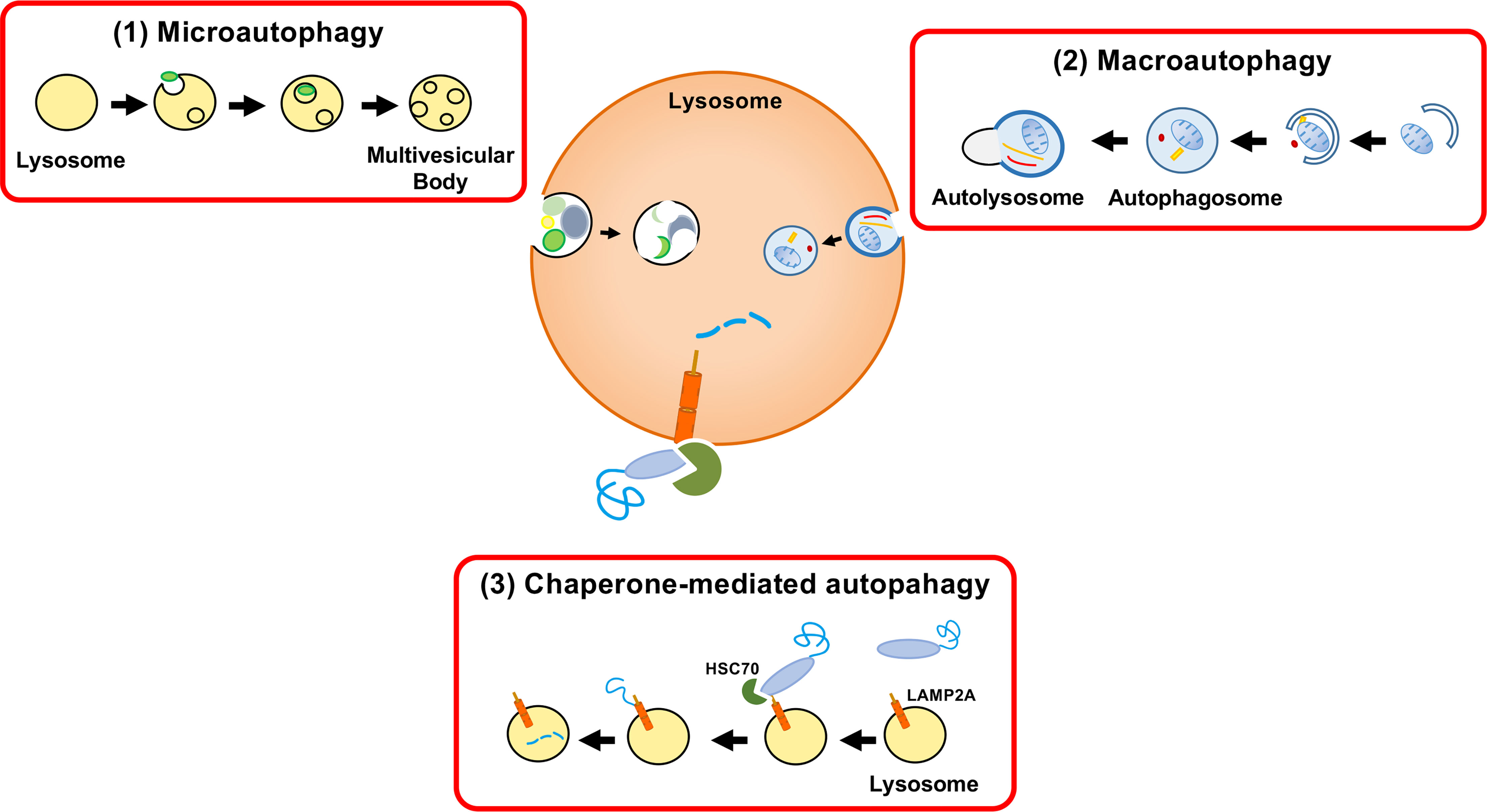
Rather, REDD1 was rapidly degraded upon activation of chaperone-mediated autophagy (CMA) and exposure to hyperglycemic conditions prevented the suppressive effect of CMA activation on REDD1 expression. Surprisingly, the suppressive effect of oxidative stress on REDD1 degradation was independent of the ubiquitin-proteasome pathway. 
Antioxidant addition to culture medium prevented both the increase in oxidative stress and REDD1 stability in cells exposed to hyperglycemic conditions. In human MIO-M1 retinal Müller cells, the rate of REDD1 degradation was reduced upon exposure to hyperglycemic conditions. When diabetic mice were administered antioxidants, neither oxidative stress nor REDD1 expression were increased in the retina. In the retina of streptozotocin-induced diabetic mice, REDD1 protein content was increased in the absence of a change in REDD1 mRNA. Herein, we investigated the hypothesis that diabetes promotes REDD1 expression by reducing the rate of REDD1 degradation. However, the mechanism responsible for increased retinal REDD1 protein content in the context of diabetes has never been established. In fact, intravitreal administration of a siRNA targeting the REDD1 mRNA has demonstrated promise for improving visual function in patients with diabetic macular edema. Upregulation of the stress response protein REDD1 (regulated in development and DNA damage response 1) has been implicated in diabetes-induced retinal defects in both preclinical models and diabetic patients.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
